Almost everyone learns in school that mitochondria are "the powerhouses of the cell." This is true, as far as it goes. But mitochondria are vastly more complex and more important than that familiar phrase suggests. They are the sites where food and oxygen are converted into the energy currency of life. They are also exquisitely sensitive to their electromagnetic environment — and they depend on melatonin, produced locally within their own membranes, to protect them from the very energy-production process they carry out. Understanding this relationship sheds new light on why the bedroom's EMF environment during sleep may matter more than most people realize.
The Electron Transport Chain: Energy Production at the Quantum Scale
Mitochondria produce ATP — adenosine triphosphate, the molecule that powers virtually every cellular process — through a mechanism called the electron transport chain (ETC). This is a series of protein complexes embedded in the inner mitochondrial membrane that pass electrons from one complex to the next in a precisely controlled sequence.
As electrons flow through the chain, they release energy that is used to pump hydrogen ions (protons) across the inner membrane, creating an electrochemical gradient. This gradient drives the final enzyme in the chain — ATP synthase — which literally spins like a molecular turbine, synthesizing ATP as protons flow back through it. It is one of the most remarkable molecular machines in biology.
The efficiency of this process depends on precise conditions. The electron transport chain operates at the quantum level — electrons must tunnel through protein complexes at specific rates, and the proton gradient must be maintained within a narrow range. Disruptions to membrane integrity, electron flow, or the surrounding electromagnetic environment can impair this process, reducing ATP output and increasing the production of reactive oxygen species (ROS) — essentially, exhaust from an engine running inefficiently.
Water Structure and the Inner Membrane
The inner mitochondrial membrane is not just a passive barrier. It is a dynamic structure whose function depends, in part, on the behavior of water at its surfaces. Gerald Pollack, a professor of bioengineering at the University of Washington, has published extensively on a phenomenon he calls exclusion zone (EZ) water — a structured, gel-like phase of water that forms at hydrophilic surfaces.
Pollack's research, published in multiple peer-reviewed journals and compiled in his book The Fourth Phase of Water (2013), demonstrates that water adjacent to certain surfaces develops a structured layer that excludes solutes, carries a negative charge, and has different physical properties from bulk water. This structured water phase is driven by radiant energy — particularly infrared wavelengths.
While the application of EZ water theory to mitochondrial function is still being developed and debated in the scientific community, the conceptual framework is intriguing: the inner mitochondrial membrane, with its vast surface area of cristae folds, is precisely the kind of hydrophilic surface where structured water would be expected to form. If the water structure at this interface contributes to the proton gradient or electron flow, then factors that disrupt water structuring — including electromagnetic interference — could potentially affect energy production.
Melatonin: The Mitochondrial Guardian
Here is where the story becomes truly remarkable. Melatonin is best known as the hormone that regulates sleep-wake cycles, secreted by the pineal gland in response to darkness. But research over the past two decades has revealed that melatonin is also produced locally within mitochondria — and this local production may be its most important function.
The electron transport chain inevitably produces some reactive oxygen species as byproducts — superoxide radicals, hydrogen peroxide, and hydroxyl radicals. These are highly reactive molecules that can damage the mitochondrial DNA, membrane lipids, and the ETC proteins themselves. Over time, this oxidative damage accumulates and is a major driver of cellular aging and dysfunction.
Melatonin acts as a free radical scavenger directly within mitochondria — right at the source of ROS production. It neutralizes superoxide and hydroxyl radicals, stimulates the production of other antioxidant enzymes (including superoxide dismutase and glutathione peroxidase), and protects mitochondrial DNA from oxidative damage. No other endogenous antioxidant operates with this combination of potency and specificity at the mitochondrial level.
This means that adequate melatonin production is not merely important for falling asleep — it is essential for protecting the very organelles that produce your cellular energy. When melatonin levels are suppressed — by artificial light, disrupted circadian rhythms, or potentially by electromagnetic interference — mitochondria lose a critical layer of protection during the hours when repair should be at its peak.
The Sleep Window: When Repair Happens
Sleep is not a passive state. It is the body's most metabolically active repair period. During deep sleep, particularly between approximately midnight and 2 AM (assuming a typical sleep schedule), the body releases a concentrated pulse of anabolic hormones:
- Growth hormone — essential for tissue repair, muscle recovery, bone maintenance, and fat metabolism. The majority of daily growth hormone secretion occurs during the first few hours of deep (NREM stage 3) sleep.
- Prolactin — supports immune function and tissue repair. Prolactin secretion rises during sleep and is suppressed by sleep disruption.
- Testosterone — peaks during sleep and is dependent on adequate sleep duration and quality. Sleep restriction studies consistently show reduced testosterone levels.
The efficacy of these hormones depends on properly functioning mitochondria. Growth hormone stimulates protein synthesis and cell division — processes that require substantial ATP. Immune function requires energy for cell proliferation and antibody production. DNA repair mechanisms need both energy (ATP) and protection (melatonin) to operate effectively.
When mitochondrial function is impaired — whether by oxidative stress, calcium dysregulation, or reduced melatonin — the entire repair cascade is compromised. You may still sleep for eight hours, but the quality of the repair work that happens during those hours may be diminished.
The EMF Connection
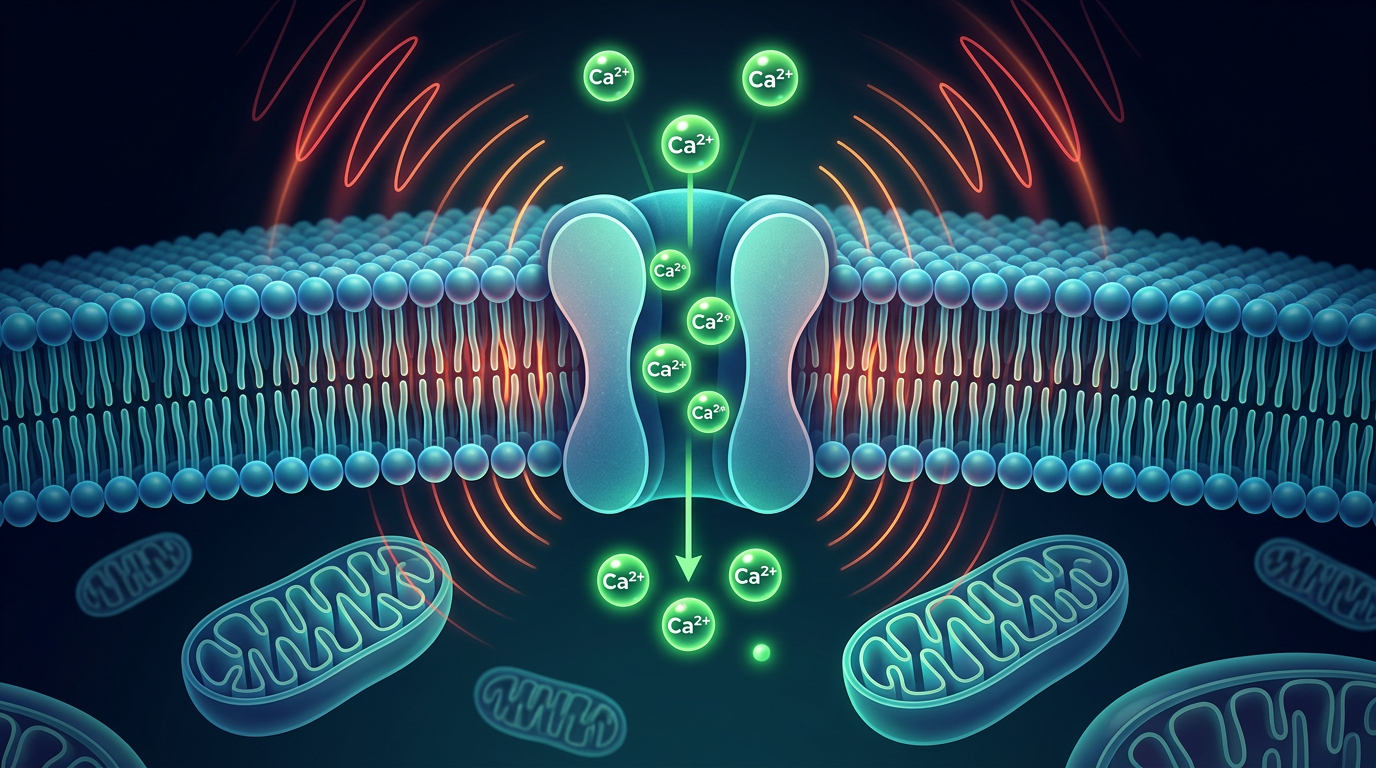
This is where the various threads converge. Research on voltage-gated calcium channels suggests that non-native EMF may trigger inappropriate calcium influx into cells. Excess intracellular calcium is a well-known mitochondrial stressor — it can depolarize the mitochondrial membrane, inhibit ATP synthase, increase ROS production, and even trigger apoptotic (cell death) pathways if severe enough.
If EMF-induced calcium dysregulation impairs mitochondrial function, the consequences compound: less efficient energy production means more ROS, more ROS means greater need for melatonin, and if melatonin production is itself suppressed by the same electromagnetic environment, the mitochondria face a double burden — increased oxidative stress with reduced antioxidant protection.
This is a hypothesis, not an established fact. The research connecting these dots is still developing, and each link in the chain has varying degrees of evidentiary support. But the biological plausibility is strong, and the precautionary logic is clear.
Why Sleep Feels Different in a Low-EMF Bedroom
Many people who reduce the EMF levels in their bedrooms — by turning off WiFi, removing wireless devices, and sometimes installing RF shielding — report that their sleep feels qualitatively different. They describe waking feeling more refreshed, experiencing fewer nighttime awakenings, and having more vivid dreams (a sign of adequate REM sleep).
These subjective reports are not proof of mechanism, but they are consistent with what the science would predict. If reducing nnEMF during sleep allows for more orderly calcium regulation, less mitochondrial stress, fuller melatonin production, and more efficient repair processes, the subjective experience of better rest would be the expected outcome.
The beauty of this intervention is its simplicity. You don't need to understand quantum tunneling in the electron transport chain to benefit from turning off your WiFi at night. You don't need a genetics degree to put your phone in airplane mode. The actions are easy; the potential benefits are rooted in fundamental cellular biology.
Key Takeaways
- Mitochondria produce ATP through the electron transport chain — a process that depends on precise electromagnetic and biochemical conditions
- Melatonin is produced locally within mitochondria and serves as their primary antioxidant defense against the ROS inherent to energy production
- Sleep is the body's peak repair period — growth hormone, prolactin, and other anabolic hormones depend on mitochondrial energy production and melatonin protection
- EMF-induced calcium dysregulation is a plausible mechanism for mitochondrial stress, potentially creating a double burden of increased ROS and reduced melatonin
- Gerald Pollack's exclusion zone water research raises questions about how electromagnetic conditions may affect water structure at the mitochondrial membrane
- Reducing nnEMF during sleep is a simple, low-cost way to support the conditions mitochondria need for optimal repair and regeneration